
Tags Archive Navigation
icon
-
Media ReleaseNovartis radioligand therapy Lutathera® FDA approved as first medicine specifically for pediatric patients with gastroenteropancreatic neuroendocrine tumors
-
Story
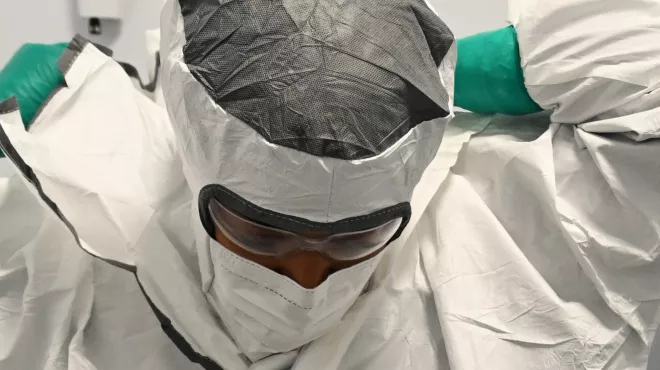 Drug delivery, service delivery, and the future of cancer care
Drug delivery, service delivery, and the future of cancer care -
Story
 Listening to the CML patient community
Listening to the CML patient community -
Media ReleaseNovartis to strengthen oncology pipeline with agreement to acquire MorphoSys AG for EUR 68 per share or an aggregate of EUR 2.7bn in cash
-
Story
 Facing the fear of breast cancer recurrence
Facing the fear of breast cancer recurrence -
Media ReleaseNovartis Lutathera® significantly reduced risk of disease progression or death by 72% as first-line treatment for patients with advanced gastroenteropancreatic neuroendocrine tumors
-
Media ReleaseNovartis Scemblix® shows superior major molecular response (MMR) rates vs. standard‐of‐care TKIs in Phase III trial for newly diagnosed patients with chronic myeloid leukemia
-
Media ReleaseNovartis expands production of Pluvicto™ with addition of its largest and most advanced radioligand therapy manufacturing facility in Indianapolis
-
Media ReleaseLatest Novartis Kisqali® NATALEE analysis reinforces 25% reduction in risk of recurrence across broad population of patients with early breast cancer; supports regulatory submissions
-
Landing PageMy Cancer. My Words.
-
Story
 Shoulder to shoulder
Shoulder to shoulder -
Interior PageOncology
Pagination
- ‹ Previous page
- 1
- 2
- 3
- 4
- 5
- 6
- 7
- …
- 15
- › Next page