Novartis Pharmaceuticals Corporation (“Novartis”), in cooperation with the U.S. Consumer Product Safety Commission (“CPSC”) and Food and Drug Administration (“FDA”), is voluntarily implementing a CPSC-approved corrective action plan for the following product packages distributed in the United States. This action is not a result of any quality or safety concerns with the medications for their intended use.
The blister cards in which these products are packaged are not child-resistant, posing a risk of harm if the tablets are swallowed by children. Products in bottles are NOT impacted.
Zofran ODT® 4mg and 8mg (ondansetron) Orally Disintegrating Tablets (ODT)
This notification applies ONLY to the NDCs and lot numbers below:
| Product Description | Package Description | NDC Number on Carton | NDC Number on Blister Pack | Lot Number | Expiration Date |
|---|---|---|---|---|---|
| Zofran ODT 4 mg | Blister packs of 30 tablets | 0078-0679-19 | 0078-0679-61 | 1657088 | Dec 2019 |
| Zofran ODT 8 mg | Blister packs of 30 tablets | 0078-0680-19 | 0078-0680-61 | 1641546 | Oct 2019 |
ENTRESTO® (sacubitril / valsartan) Tablets in 100 Count Hospital Unit Dose Blister Packages
This notification applies ONLY to the NDCs and lot numbers below:
| Product Description | NDC Number on Carton | NDC Number on Blister Card | Lot Number | Expiration Date |
|---|---|---|---|---|
| Entresto FCT 24mg/26mg HUD | 0078-0659-35 | 0078-0659-61 |
FX000005 |
Apr 2020 |
| Entresto FCT 49mg/51mg HUD | 0078-0777-35 | 0078-0777-61 |
FX000001 |
Dec 2019 |
| Entresto FCT 97mg/103mg HUD | 0078-0696-35 | 0078-0696-61 |
FX000002 |
Mar 2020 |
These blister packs were developed solely for use in institutions (i.e., hospitals). However, we learned that some of these institutional packs were sent to retail pharmacies, indicating that they may have been dispensed for in-home use.
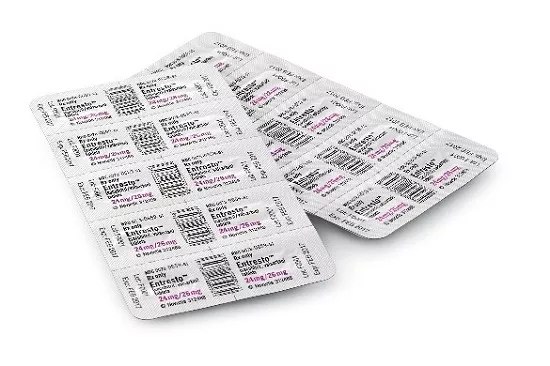
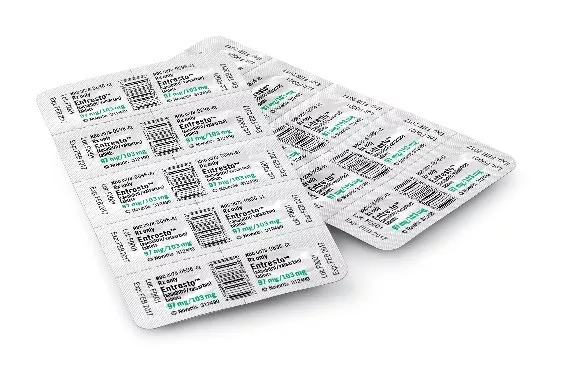
REPRESENTATIVE PHOTOS:
These images show the type of packaging involved in the voluntary recall. Products in bottles are NOT impacted. NDC numbers, lot numbers and expiration dates in the following example images should be disregarded. They are NOT those involved in the recall. Please refer to the table of affected lots above.

|

|
| Zofran ODT 4mg | Zofran ODT 4mg |

|

|
| Zofran ODT 8mg | Zofran ODT 8mg |
|

|
| Entresto FCT 24mg/26mg HUD | Entresto FCT 49mg/51mg HUD |
|
|
|
Entresto FCT 97mg/103mg HUD |
PATIENT ACTION:
Anyone having the packages listed above should call Novartis Pharmaceuticals Corporation at 888-NOW-NOVA (888-669-6682) from 8 a.m. to 8 p.m. ET Monday through Friday and 9 a.m. to 6 p.m. ET Saturday and Sunday to order a child-resistant pouch in which to store the blister packs within your possession.
The quality of these products has not been compromised, so patients should continue taking their medicine as directed by their physician. However, due to the risk of harm to children, immediately secure this medicine so that it is out of the sight and reach of children.
Instructions for using the child-resistant pouch are on the pouches themselves, and the following video demonstrates their use.
Further information about this recall is available by calling Novartis at 888-NOW-NOVA (888-669-6682).
To learn more about the Sandoz products affected by this recall and requiring corrective action, please click here to be redirected to the Sandoz website.
Any medical related inquiries should be directed to Novartis Pharmaceuticals Corporation at 888-NOW-NOVA (888-669-6682). Please report any adverse events by calling Novartis at the same phone number, or by e-mailing usdrugsafety.operations@novartis.com. Adverse events can also be reported to the FDA online at www.fda.gov/medwatch/report.htm.
Click the images below for the related retail posters: